Θυμηθείτε πως...
....γίνεται η εφαρμογή της δοκιμής Schirmer και της δοκιμής της φλουορεσκεΐνης
> Εισαγωγή
Η δοκιμή Schirmer και η δοκιμή της φλουορεσκεΐνης είναι δυο απλές διαγνωστικές δοκιμές που εφαρμόζονται πολύ συχνά για τη διάγνωση ορισμένων οφθαλμολογικών παθήσεων στα ζώα συντροφιάς. Η σωστή εκτέλεση των δοκιμών αυτών ελαχιστοποιεί τις πιθανότητες λανθασμένης ερμηνείας των αποτελεσμάτων τους, και οδηγεί στην ασφαλή διάγνωση.
> Η δοκιμή Schirmer
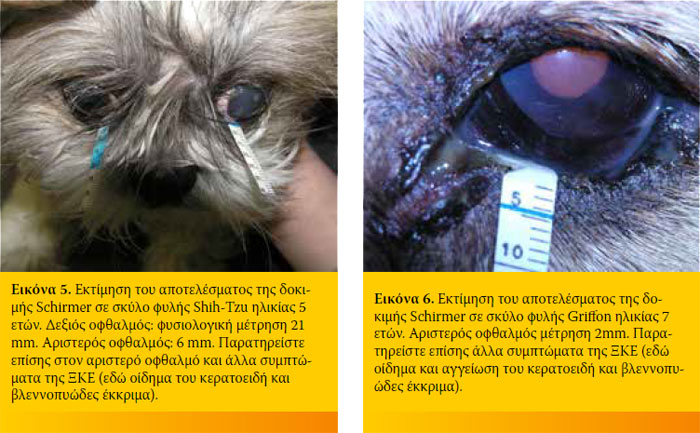
Με τη δοκιμή Schirmer ελέγχεται η παραγωγή της υδάτινης φάσης των δακρύων. Κατά συνέπεια χρησιμοποιείται για τη διάγνωση της ξηρής κερατοεπιπεφυκίτιδας (ΞΚΕ) και ειδικότερα της ΞΚΕ ποσοτικού τύπου. Η διάγνωση της ποιοτικού τύπου ΞΚΕ γίνεται με άλλες εξετάσεις. Στην κλινική πράξη εφαρμόζεται η δοκιμή Schirmer I η οποία γίνεται χωρίς τοπική αναισθησία και η οποία υπολογίζει τόσο τη βασική όσο και την αντανακλαστική έκκριση των δακρύων. Οι φυσιολογικές τιμές στη δοκιμή Schirmer κυμαίνονται από 18,64 ± 4,47 mm /min έως 23,90 ± 5,12 mm /min στον ενήλικο σκύλο και από 14,3 ± 4,7 mm/min έως 16,92 ± 5,73 mm/min στην ενήλικη γάτα στην οποία πάντως η ΞΚΕ είναι σπανιότερη. Στο σκύλο μέτρηση μικρότερη των 5 mm δηλώνει σοβαρής μορφής ΞΚΕ. Μέτρηση 5-10 mm δηλώνει ΞΚΕ ενώ 10-15mm θέτει υπόνοια για τη νόσο και πρέπει να συνεκτιμάται με τα άλλα κλινικά συμπτώματα.
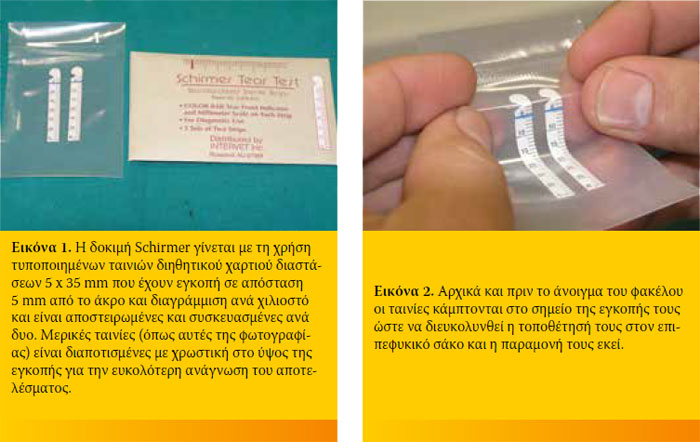

Η δοκιμή Schirmer πρέπει να γίνεται νωρίς στην οφθαλμιατρική εξέταση για να αποφεύγεται η διέγερση της αντανακλαστικής έκκρισης των δακρύων λόγω των χειρισμών και πριν την τοπική χρήση άλλων διαγνωστικών διαλυμάτων. Κατά τις επανεξετάσεις έλεγχου της ΞΚΕ δεν θα πρέπει να χορηγείται θεραπεία τις ημέρες των μετρήσεων. Πριν από την εξέταση η βλέννα και το έκκριμα που πιθανά υπάρχει στα βλέφαρα και τον επιπεφυκικό σάκο, απομακρύνεται ήπια αλλά χωρίς σχολαστικότητα και χωρίς έκπλυση.
> Η δοκιμή της φλουορεσκεΐνης
Η συχνότερη ένδειξη για την τοπική χρήση της φλουορεσκεΐνης, που θα αναφερθεί και εδώ, είναι για την ανίχνευση του έλκους του κερατοειδή χιτώνα. Η φλουορεσκεΐνη χρησιμοποιείται επίσης για την ανίχνευση διαφυγής υδατοειδούς υγρού από τον πρόσθιο θάλαμο (δοκιμή Seidel), για τη διάγνωση της ποιοτικού χαρακτήρα ΞΚΕ (δοκιμή του χρόνου ρήξης της προκεράτιας δακρυϊκής στοιβάδας) και τέλος για τον έλεγχο της διαβατότητας της δακρυϊκής συσκευής.
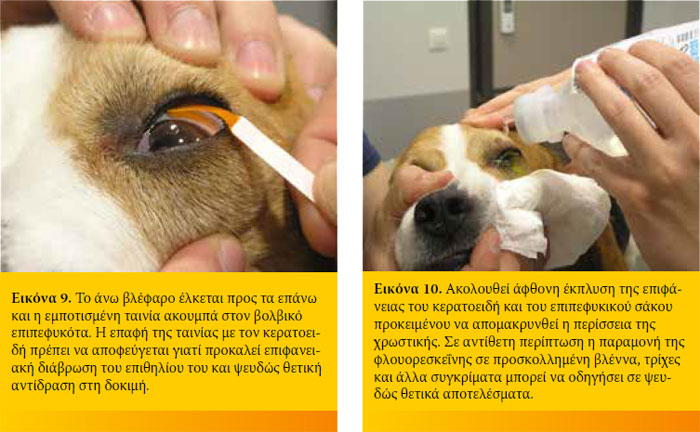
Η φλουορεσκεΐνη παρουσιάζει το μέγιστο της φασματικής απορρόφησης του φωτός στα 490 nm (μπλε φως) και έχει την ιδιότητα να μετατρέπει το 100% του απορροφούμενου φωτός στα 520 nm (πράσινο φως). Για το λόγο αυτό η ανίχνευσή της είναι ευκολότερη όταν φωτίζεται με μπλε φως.
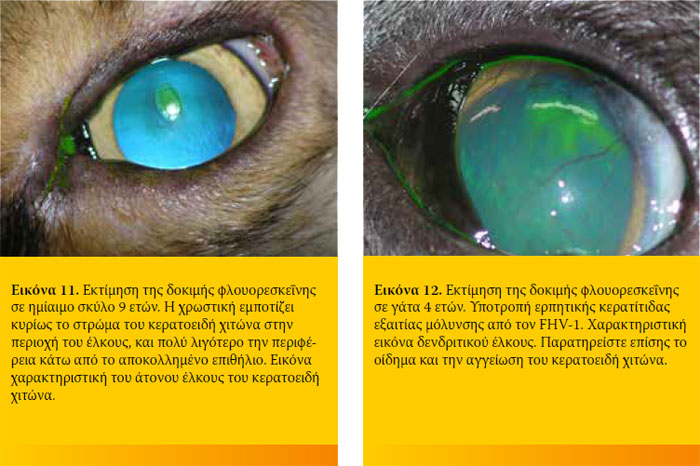
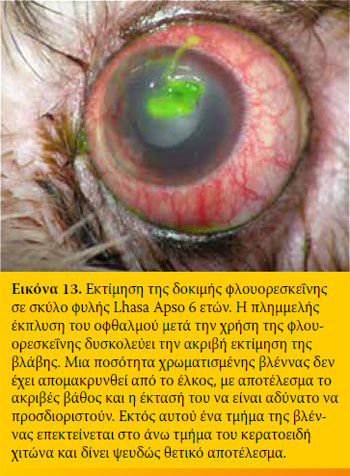
Η φλουορεσκεΐνη είναι ισχυρά λιπόφοβη και υδρόφιλη ουσία. Έτσι δεν συγκρατείται από το υγιές επιθήλιο του κερατοειδή λόγω του λίπους που περιέχεται στις κυτταρικές μεμβράνες του. Σε περίπτωση βλάβης του επιθηλίου όμως, η φλουορεσκεΐνη διηθεί το στρώμα του κερατοειδή προσδίδοντάς του το χαρακτηριστικό πράσινο χρώμα. Η φλουορεσκεΐνη δεν απορροφάται από τη μεμβράνη του Descemet. Για το λόγο αυτό στα προδεσκεμετικά έλκη βάφει μόνον τη περιφέρεια και όχι τον πυθμένα τους.
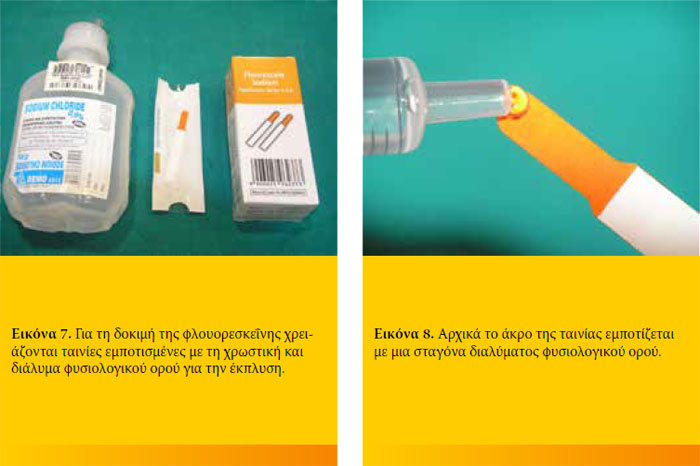
Στο εμπόριο η φλουορεσκεΐνη κυκλοφορεί είτε σε μορφή διαλύματος 2% σε μορφή φιαλιδίων μιας δόσης, είτε εμποτισμένη σε χάρτινες ταινίες μιας χρήσης σε στείρα συσκευασία. Η δεύτερη μορφή είναι πιο εύκολο να βρεθεί στην Ελλάδα.Τα διαλύματα πολλαπλής χρήσης πρέπει να αποφεύγονται γιατί θεωρούνται επιμολυσμένα. Ο έλεγχος συγκράτησης της φλουορεσκεΐνης μπορεί να γίνει και με γυμνό οφθαλμό. Για την ασφαλέστερη όμως αξιολόγηση της δοκιμής καλό είναι να γίνεται σε περιβάλλον χαμηλού φωτισμού με φως μπλε χρώματος και χρήση κάποιου μεγεθυντικού μέσου.
> Bιβλιογραφία
1. Featherstone HJ, Heinrich CL. Ophthalmic examination and diagnostics Part 1. In : Veterinary Ophthalmology. Gelatt KN, Gilger BC, Kern TJ (ed). 5th edn. John Wiley & Sons, Inc: 2013, pp. 533-613




