> Abstract
Hip dysplasia in dogs is a multifactorial disease caused by hereditary and environmental factors. Most commonly seen among very young giant and chondrodystrophic breeds, it manifests as an abnormal development of the round ligament of the femoral head that leads to coxofemoral joint instability and (sub)luxation. The disease presents as hind limb lameness. In the young animal, this is due to pain caused by hyperextension of the soft tissues of the hip joint, whereas in adults it is caused by osteoarthritis resulting from the degenerative development of the disease. In addition to radiological examination, diagnosis is based on clinical signs and specific clinical trials in both the awake animal and the animal under deep sedation or/and general anaesthesia. Radiologic imaging requires several views, and findings are evaluated according to the animal’s age, stage of disease and clinical signs. The need for early assessment of hip dysplasia resulted in Prassinos N.N. the establishment of different classification systems according to radiological findings.
> Introduction
A very descriptive definition of hip dysplasia (HD) was given by Henricson et al. (1966): “Hip dysplasia is a disease that stems from a varying degree of laxity of the hip joint, permitting subluxation during early life, giving rise to varying degrees of shallow acetabulum and flattening of the femoral head, finally inevitably leading to osteoarthritis”.1
Numerous methods have been described for the diagnosis of HD in dogs, a large number of which may prove confusing for the clinical practitioner. This study aims to present the aetiology and pathogenesis of HD, as well as the main diagnostic procedure.
> Aetiopathogenesis
The true cause of HD remains unclear. However, after 75 years of research, it is widely acceptable that HD reflects the interaction of multiple genes with environmental influences, although the exact mechanism of the gene expression is still unknown.2
At birth, the hip joints are normal and they are thought to continue normal development if complete congruity between the femoral head and the acetabulum is maintained. 3-13 HD can be best considered as the result of the «duel» between the forces applied to the hip during weight-bearing of the limb and the rapidity of cartilage development to bone. This means that if the growing cartilage of the coxofemoral joint turns into a normal morphology bone before the effects of joint forces, dysplasia can probably be avoided, otherwise HD is inevitable.14,15
Two endogenous factors are suspected in the early laxity of the coxofemoral joint. The first refers to the abnormal production of collagen fibres in the articular capsule and the round ligament of the femur16 and the second to the irregular endochondral ossification of the bones that constitute the coxofemoral joint, which leads to contact incongruity of the articular surface.14-16
During development of the hip, the earliest dysplastic joint changes are observed at 30 days of age and are related to the round ligament of the femoral head, which at this stage is primarily responsible for maintaining hip joint stability. More specifically, an oedematous ligament is observed, as well as torn fibres and capillary haemorrhage at the tearing sites. Increased volume of the ligament of the femoral head and increased synovial fluid volume have been considered the earliest findings of canine hip dysplasia. In addition, during the first two weeks of the animal’s life, the short round ligament begins to lengthen. It has been suggested that in dysplastic dogs, it is this excessive lengthening that permits lateral subluxation of the adult hip joint.3-13 It is also known that there is more than one ossification centre in the acetabulum of a young animal, with that of the dorsal rim normally detaining the junction of the acetabulum with the iliac bone. In dogs with HD, this delay is even longer.14
Certain environmental factors that do not cause the disease but can affect its appearance and development at varying degrees and can be modified by the owner after an early indication of a veterinarian include the following:
a) The body weight of the puppy and adult dog, and mostly those breeds whose body weight increases rapidly during the first months of the animal’s life.2
b) Increased amounts of calcium and vitamin D with food or nutrition supplements since they cause the endochondral ossification process to delay.15,17,18
c) Excessive exercise of the animal at a young age, as well as it residing in places with slippery floors.2
As many authors suggest, the rapid increase of body mass in large dog breeds is either genetically predefined or due to excessive nutrition rich in high calories and ingredients which has a crucial effect on the hip joint, thereby contributing to the premature appearance of dysplasia which predisposes to more severe development. On the contrary, a slower growth rate provides the joint with the opportunity to develop normally.14
From a bio-mechanical point of view, HD could be attributed to the asynchronous growth between the muscle mass and the skeleton which evolves faster. In particular, inability or failure of the hip muscles to obtain sufficient length, volume and functionality in a time relative to skeleton development will greatly affect hip stability, triggering a sequence of events which will lead to dysplasia and osteoarthritis. Notably, the stability of the hip depends greatly on muscle strength during motion, as well as at standing position.14,19
The first radiographic signs of canine HD that can be seen as early as at 7 weeks of age are subluxation of the femoral head and underdevelopment of the craniodorsal acetabular rim. At this time, the joint capsule is stretched but not otherwise structurally altered, and the ligament of the femoral head is lengthened.9,10
At the age of 2-3 months, the degree of subluxation increases and the joint capsule suffers further stretching, thickness and inflammation. This inflammation coexists with an increased production of prostaglandins by the cells of the synovium, which causes vasodilation in the articular capsule and pain. The synovial fluid, produced by the cells of the synovial membrane and extravasation of plasma, is significantly increased in quantity, while its viscosity is reduced because of greater plasma extravasation, which ultimately does not serve the lubrication needs of the articular cartilage. The femoral head is laterally displaced, and in more severe cases cranially in relation to the acetabulum. The articular cartilage begins to erode and roughen, mainly on the dorsal surface of the femoral head, especially at the contact site with the rim of the acetabulum.20
In a healthy, congruent hip joint, forces during weight-bearing are distributed across the entire cartilaginous surface of the acetabulum and the vicinity muscles, which helps to balance those forces. On the contrary, the head of a dysplastic hip due to subluxation is tangent only to a small part of the dorsal acetabular rim, which receives much greater forces than it can withstand. The latter results in microfructures and progressive destruction of the articular cartilage of the dorsal acetabular rim, inflammation of the coxofemoral joint and reactive periarticular fibration, progressive deformation of the articular surfaces, remodelling of the dorsal acetabular rim, and osteophyte production. The consequence of all the above is hip osteoarthritis.21
> Diagnostic approach
HD may be suspected in a dog of any age belonging to those breeds with increased predisposition that presents with the symptoms described above.
A full history of the patient is initially obtained, in addition to physical, orthopedic and neurological examination, so as to limit the origin of the clinical symptoms to the hip joint and exclude other orthopedic or neurologic conditions that may present in a similar way to HD. Several dog breeds that develop HD, are considerably predisposed to hind limb lameness of different aetiology. These pathologic conditions need to be recognised and differentiated from HD in order to avoid misdiagnosis and possible unsuccessful treatment. This is of particular importance in the case of coexistent HD that is not responsible for hind limb lameness. With the exception of injury, other causes in young dogs include patella luxation, panosteitis, aseptic necrosis of the femoral head, osteochondritis and on rare occasions, hypertrophic osteodystrophy, while in adult animals, causative factors are cranial cruciate ligament rupture, patella luxation, degenerative lumbosacral stenosis, discospondylitis, intervertebral disc protrusion, polyarthritis and bone tumours.2,20,22
Almost all diagnostic methods used for humans have also been applied to dogs: arthrography, arthroscopy, ultrasonography, thermography, scintigraphy, and in special cases, computed tomography and magnetic resonance imaging. Nevertheless, complete physical and radiological examination of the hips, are usually sufficient for the confirmation of the disease.23
> Epidemiology
Theoretically HD can be presented in dogs of any breed, although those mostly affected are large and giant breeds, as well as chondrodystrophic breeds regardless of their size. Consequently, according to the OFA (Orthopedic Foundation for Animals) database (www.offa.org), dogs belonging to breeds such as Newfoundland, Saint Bernard, Old English sheepdog, Rottweiler, German Shepherd Dog, Samoyed, Golden Retriever, Alaskan Malamute, Labrador Retriever, and also Pug, have the highest incidence of the disease.
> History – Clinical presentation
The clinical symptoms of HD vary and are characterised as ranging from mild discomfort to severe acute or chronic pain. The disease evolves progressively24 and clinical manifestation differs in regard to the dog’s age. Two age groups can be identified: young (4-16 months) and adult (>16 months).9
Regarding young dogs, clinical symptoms most often appear at 4-12 months of age; they occur suddenly and are characterised by unilateral or bilateral hind limb lameness. The animals exhibit difficulty in rising; they prefer to sit and are reluctant to run, jump, climb up and down stairs and even walk, while in general they resent any motor activity. Their gait is of shorter width, while at faster gait, especially when running and climbing stairs, they move both hind limbs simultaneously (bunny-hopping). Dysplastic dogs show hind limb stiffness after a long period of inactivity or when inactivity is followed by a copious activity.2 The aforementioned clinical presentation is attributed to pain, which is mainly caused by the changes occurring to the anatomical features of the hip joint region due to extreme laxity of the joint, as well as the abnormal motility of the subluxated femoral head.9
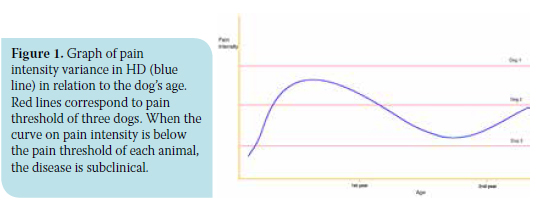
Some affected puppies may never exhibit any clinical symptoms and the disease could remain subclinical, taking into consideration that in such cases pain severity does not surpass the pain threshold (Figure 1). On the other hand, other puppies exhibit notably severe pain symptoms, which in many cases disappear within the following months; hence, by the age of 12-15 months, the puppies are relatively asymptomatic, probably because of reactive periarticular fibration which seems to stabilize the joint. In the rest of the animals, pain not only remains, but progressively worsens and accompanies them in the second year of their lives.25,26
As concerns the adult group which constitutes the second phase of the disease, symptoms may appear at any age. Onset is mostly progressive and rarely sudden. The occurring symptomatology is similar to that of hip osteoarthritis.2
> Clinical examination
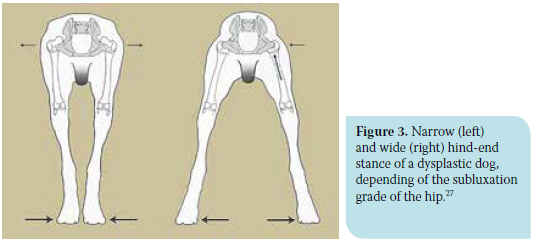
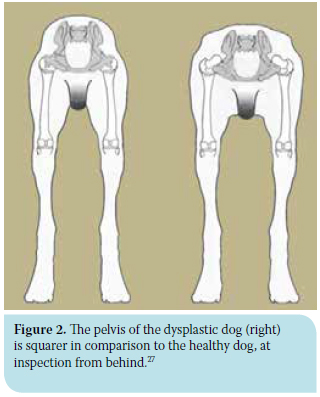 Visual inspection should be performed in the standing as well as the moving position. In standing position, dogs with severe HD display permanent subluxation of the femoral heads resulting in protrusion of the greater trochanters dorsally and laterally. Subluxation may also make the dog appear lower and wider at the hind end, which tends to become exaggerated in the case of muscle atrophy. Thus, at inspection of the animal from behind, the pelvis region seems more «squared» (Figure 2).2,27 Depending on the stage of the disease, affected animals may exhibit a wide or narrow hind-end stance (Figure 3). At first, the animals have a wide hind-end stance (limb abduction) in trying to maintain the femoral heads in the cavity of each acetabulum. As time passes and subluxation aggravates, the femoral head is increasingly supported by the thickened articular capsule, rather than the acetabulum. As a result, abduction of the limbs is reduced since due to subluxation, the strong and painful contraction of the adductor muscles, and especially the pectineus, triggers forces that act on the already inflamed and stretched articular capsule. Thus, the animal adopts narrow-end stance (limb adduction) in order to reduce the intensity of pain.27
Visual inspection should be performed in the standing as well as the moving position. In standing position, dogs with severe HD display permanent subluxation of the femoral heads resulting in protrusion of the greater trochanters dorsally and laterally. Subluxation may also make the dog appear lower and wider at the hind end, which tends to become exaggerated in the case of muscle atrophy. Thus, at inspection of the animal from behind, the pelvis region seems more «squared» (Figure 2).2,27 Depending on the stage of the disease, affected animals may exhibit a wide or narrow hind-end stance (Figure 3). At first, the animals have a wide hind-end stance (limb abduction) in trying to maintain the femoral heads in the cavity of each acetabulum. As time passes and subluxation aggravates, the femoral head is increasingly supported by the thickened articular capsule, rather than the acetabulum. As a result, abduction of the limbs is reduced since due to subluxation, the strong and painful contraction of the adductor muscles, and especially the pectineus, triggers forces that act on the already inflamed and stretched articular capsule. Thus, the animal adopts narrow-end stance (limb adduction) in order to reduce the intensity of pain.27
When the gait of a dysplastic dog is observed, a swing of the caudal body part may be noted, perhaps because the animal attempts to reduce the pain by shortening its gait. The hind limbs seem stiff, while the stifle and tarsus are relatively extended, probably due to the animal’s attempt to shift its weight onto the front limbs. The «bunny hopping» gait is a recurring symptom of HD and indicates the animal’s attempt to reduce the pain, since in order to move its body forward, the forces acting on the hips during weight-bearing are evenly distributed in both hind limbs.2
Palpation of the hind limbs and especially the hips should be performed in two phases: the first on an awake animal and the second under sedation or preferably under general anaesthesia. In the first phase, which in the case of large breed dogs should be performed on the floor, muscle atrophy of the hind limbs is first assessed, followed by passive movements of flexion, extension, external rotation and abduction of the hips to test for crepitation and pain which become more obvious at extension of the joint, as well as to assess the range of motion of the coxofemoral joint which clearly decreases in animals with HD. In the second phase, specific semiquantitative palpation manoeuvres are performed, namely the Ortolani test and the calculation of the angle of Ortolani and Barlow sign, as well as the Bardens test, which give very useful information concerning hip laxity in young animals.28-31
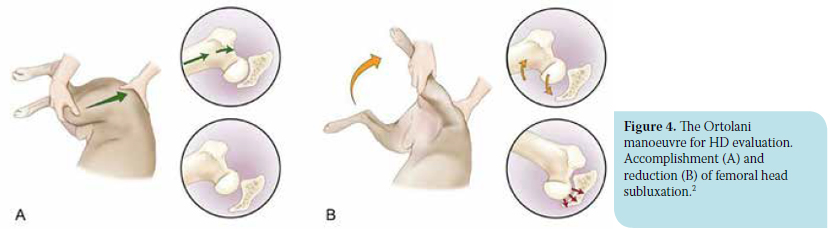
In order to perform the Ortolani test, the animal is placed in lateral recumbency (some prefer dorsal recumbency), with the limb to be examined facing upwards. For examination of the right hip, the examiner stands behind the animal and brings the right hind limb parallel to the examination table and perpendicular to the sagittal plane of the animal (spine). Using the right hand, the examiner holds the distal aspect of the stifle, while the palm of the left hand is placed on the dorsal side of the pelvis and spine to provide stabilization. The first part of the manoeuvre requires the limb in adduction; using the right hand, the examiner places medium force on the stifle joint directed through the femur toward the dorsum of the dog, while the other hand resists in order to avoid animal displacement. This force causes dorsal subluxation of the femoral head in dogs with hip laxity. (Figure 4a).2
For the second part of the Ortolani manoeuvre, the hip is gradually abducted, maintaining the intensity of the force initially applied to the stifle, until a characteristic “click” is heard and/or palpated as abrupt reduction of the hip occurs. This sound is interpreted as the positive Ortolani sign and suggests hip laxity (Figure 4b). At the moment of sound perception, the angle between the femur and the horizontal plane is defined as subluxation reduction angle. It is calculated with a goniometer or more easily with a “smart” mobile phone and the appropriate programme, such as the free application iHandy Level (iHandySoft Inc.). Subsequently, the hip is gradually adducted anew while also maintaining unabated the intensity of the initial force on the stifle until subluxation is caused again. This position is characterised as the Barlow sign and the angle between the femur and the horizontal plane is defined as the angle of subluxation and is likewise calculated.2,27,29,30
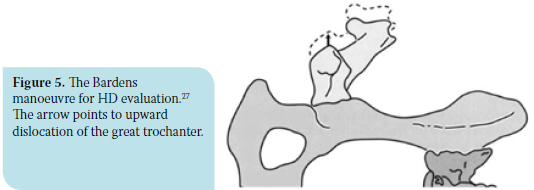
The Bardens manoeuvre is performed with the animal and examiner in the same position as the previous procedure. Using the right hand, the examiner holds the femoral diaphysis and applies force perpendicular to the sagittal plane and upwards, while the palm of the free hand is placed on the major trochanter of the same side. Major trochanter displacement upwards greater than 6 mm is interpreted as abnormal and suggests laxity of the coxofemoral joint. (Figure 5).31,32
In adult animals with HD, the result of the aforementioned tests is rarely positive, mainly due to periarticular fibrosis, remodelling of the dorsal acetabular rim, or the presence of a shallow acetabulum. Moreover, in cases of already existing subluxation due to HD, the results of these manoeuvres are negative.2
Although a positive Ortolani sign indicates laxity of the coxofemoral joint, it does not always prejudge the future clinical signs of osteoarthritis, especially when the subluxation reduction angle is small (<20ο). On the contrary, absence of the Ortolani sign does not guarantee that those animals will have normal hips during their life; in fact, they may have already developed osteoarthritic changes. Consequently, these subjective manoeuvres should be considered in relation to the results of the general examination and mainly radiographs, before final diagnosis and therapeutic plan is established.2
> Radiographic examination
Radiographic examination is a very important part of the diagnostic approach of HD in dogs and is performed with the animal under general anaesthesia. Radiographic findings are assessed according to the animal’s age, stage of the disease, clinical signs, and also the experience of the examiner. Various radiographic views are used, each of which has its advantages and disadvantages:
a) Ventrodorsal hip-extended radiograph of pelvis
b) Frog leg ventrodorsal radiograph of pelvis
c) Dorsoventral projection of the dorsal acetabular rim
d) Lateral projection of the pelvis
e) Compression ventrodorsal radiograph of the pelvis
f ) Distraction ventrodorsal radiograph of the pelvis
Proper animal positioning is essential during all of the aforementioned projections, in order to avoid false conclusions during evaluation of the radiographs.
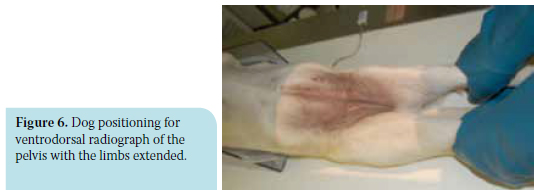
The ventrodorsal hip-extended radiograph of pelvis requires the animal to be placed in dorsal recumbency and held in that position with the use of sand bags or a special V-shaped device. Hind limbs are held by the tarsus and pulled caudally, while femurs are held parallel to each other and the spine, and slightly rotated inward until the patella projects medially in the trochlea (Figure 6). The pulling force applied for hind limb extension is determined by the ability of muscles to stretch. The most distal to the tarsus part of the hind limb should be 5-7.5 cm far from the table for small dog breeds and 10-12.5 cm for large breeds. The projected area should include the area formed by the iliac wings down to the stifle joint. The x-ray beam should be centred on the middle point of the straight line between the hips. However, providing positioning of the animal is appropriate, depiction of abnormalities in HD is not significantly affected if the beam is centred more forward or backward.23
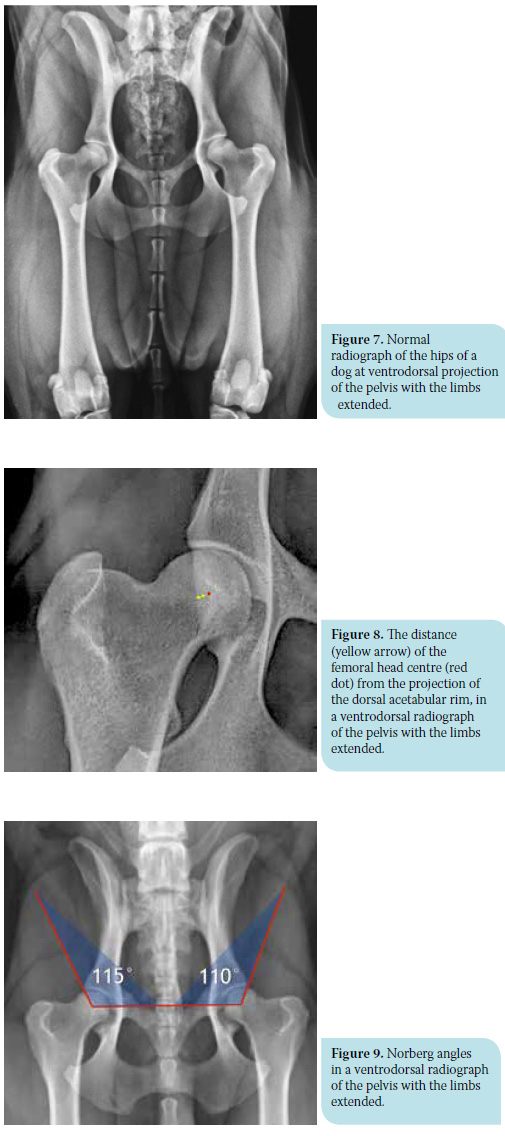 For a radiograph to be considered as correct during imaging of the hips, there needs to be adequate inward rotation of the hind limbs, the pelvis should not be rotated while at the same time, the distal part of the limbs should have the proper distance from the table (Figure 7). More specifically, if the legs are not properly rotated inwards, the angle formed by the two straight lines running through the middle level of the diaphysis and the femoral neck (anatomical axis) projects more obtuse and the limb appears valgus. This false position incorrectly suggests that the concave of the acetabulum contains a larger part of the femoral head, which fictitiously appears greater as the distance between the patella and the median projection line of the femur at the level of the trochlea increases.23 Rotation of the pelvis is indicated by the asymmetric projection of its two halves. In particular, the obturator foramen width, the shape of the iliac crest and the distance between the transverse process of the last lumbar vertebrae and the cranial rim of the iliac crest all differ between the two pelvic halves. Moreover, the sacroiliac joints are asymmetric, the penile bone does not project in a central position and finally, if the straight line between the projectional image of the spinal processes of the lumbar and sacral vertebrae is extended caudally, it does not cross the pubic symphysis. The obturator foramen width and the iliac crest shape are of greater importance when checking symmetry of the pelvis. In the case of asymmetry, the part of the pelvis where the obturator foramen seems wider and the iliac crest thinner has been elevated and turned towards the x-ray beam.23,33 As a result of this asymmetry, the acetabulum on this side appears normally shaped and deep, incorrectly covering a greater part of the femoral head,33 as opposed to the acetabulum of the other side which appears more shallow.23
For a radiograph to be considered as correct during imaging of the hips, there needs to be adequate inward rotation of the hind limbs, the pelvis should not be rotated while at the same time, the distal part of the limbs should have the proper distance from the table (Figure 7). More specifically, if the legs are not properly rotated inwards, the angle formed by the two straight lines running through the middle level of the diaphysis and the femoral neck (anatomical axis) projects more obtuse and the limb appears valgus. This false position incorrectly suggests that the concave of the acetabulum contains a larger part of the femoral head, which fictitiously appears greater as the distance between the patella and the median projection line of the femur at the level of the trochlea increases.23 Rotation of the pelvis is indicated by the asymmetric projection of its two halves. In particular, the obturator foramen width, the shape of the iliac crest and the distance between the transverse process of the last lumbar vertebrae and the cranial rim of the iliac crest all differ between the two pelvic halves. Moreover, the sacroiliac joints are asymmetric, the penile bone does not project in a central position and finally, if the straight line between the projectional image of the spinal processes of the lumbar and sacral vertebrae is extended caudally, it does not cross the pubic symphysis. The obturator foramen width and the iliac crest shape are of greater importance when checking symmetry of the pelvis. In the case of asymmetry, the part of the pelvis where the obturator foramen seems wider and the iliac crest thinner has been elevated and turned towards the x-ray beam.23,33 As a result of this asymmetry, the acetabulum on this side appears normally shaped and deep, incorrectly covering a greater part of the femoral head,33 as opposed to the acetabulum of the other side which appears more shallow.23
The process of evaluation and assessment of this projection and the kind of radiological findings differ, depending on the animal’s age. Consequently, this projection is used in adult animals to estimate hip osteoarthritis, whereas in those that are young and in whom osteoarthritis lesions have not yet been established, it is used to estimate hip joint laxity.34,35 In fact, the latter is estimated by the degree of coverage of the femoral head by the projection of the acetabulum, in which case the distance between the centre of the femoral head and the dorsal acetabular rim is calculated (Figure 8). Normally, the centre of the femoral head should be located medially to the dorsal acetabular rim and should be at least 50% covered by the acetabulum. Smaller quantum of coverage equals a loose joint.36
For each hip, the Norberg angle (NA) is formed by the straight line between the two femoral heads and the line drawn from the centre of each head to the cranial rim of the ipsilateral acetabulum (Figure 9). Normal hips are considered those where the Norberg angle is greater than 105°, although in some chondrodystrophic breeds this angle is generally smaller.37 This angle is of little diagnostic value in animals younger than five months because the radiolucent cartilage of the dorsal acetabular rim in such animals has not yet been ossified, which could lead to a false calculation.36
With regard to mainly mature animals, apart from the aforementioned calculations with which the stability of the hip is estimated in combination with the development of the dorsal acetabular rim, secondary degenerative changes observed in the acetabulum, as well as the head and neck of the femur, are also of interest:
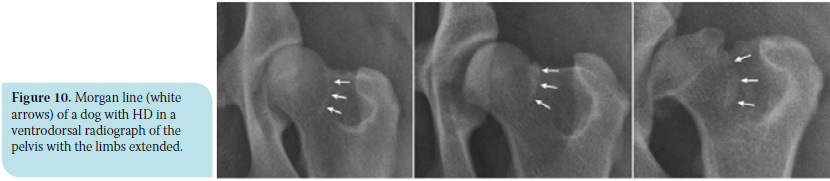
a) The Morgan line (caudolateral curvilinear osteophyte – CCO) is a well-defined linear density at the caudolateral surface of of the femoral neck, indicating osteophyte development at this position (Figure 10). The Morgan line is a constant radiographic finding in unstable hips caused by bone stimulation in the places where the synovial bursa adheres due to increased motility of the dorsal part of the femur.
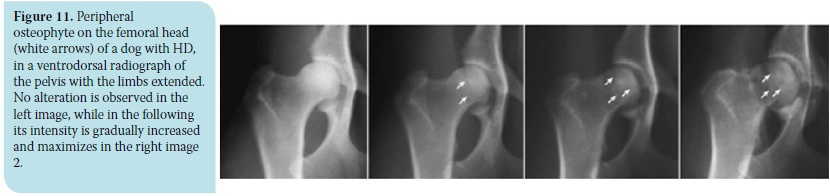
b) The circumferential femoral head osteophyte (CFHO) denotes osteophyte development at the articular margin of the femoral head and appears as a radiopaque (white) line that may or may not extend completely around the femoral head (Figure 11).2
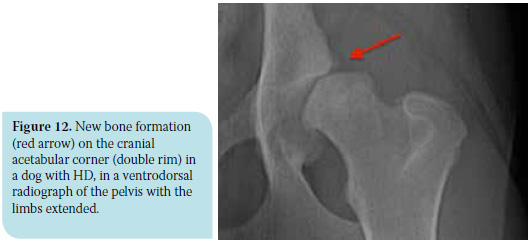
c) In an unstable joint, the craniolateral rim (cranial angle) of the acetabulum bears more weight as a result of which new bone tissue is formatted giving the impression of a double rim (Figure 12). This finding is early evidence of osteoarthritis.
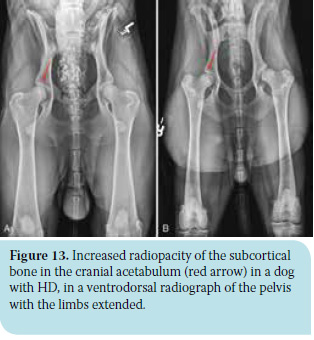 d) The subchondral bone of the cranial part of the acetabulum normally appears as a curve with normally increasing density since the pressure placed on it by the femoral head is evenly distributed during weight-bearing of the limb. In the case of a loose hip with femoral head subluxation, the pressure applied to the subchondral bone is greater and unevenly distributed, resulting in a further increase in radiopacity and loss of its consistency (Figure 13). This variable is of great diagnostic value, even when the midjoint space is narrow.23
d) The subchondral bone of the cranial part of the acetabulum normally appears as a curve with normally increasing density since the pressure placed on it by the femoral head is evenly distributed during weight-bearing of the limb. In the case of a loose hip with femoral head subluxation, the pressure applied to the subchondral bone is greater and unevenly distributed, resulting in a further increase in radiopacity and loss of its consistency (Figure 13). This variable is of great diagnostic value, even when the midjoint space is narrow.23
e) Periarticular osteophyte formation as degenerative disease evolves.
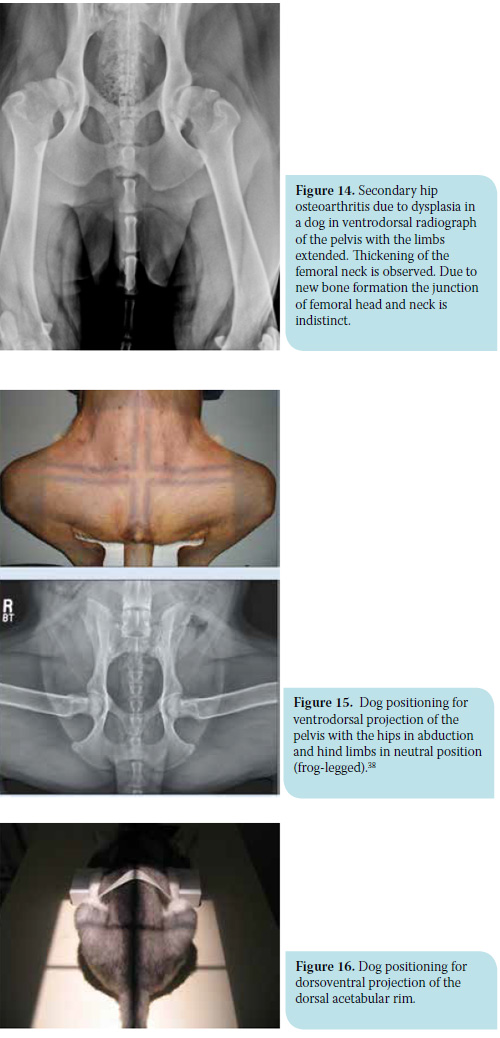
f ) Changes in the morphology of the anatomical features of the area. The acetabulum, in particular, becomes shallow and its semicircular shape appears rather semi-elliptical. The femoral head loses its spherical shape, becomes broader and sometimes resembles a mushroom. At the same time, the borders between femoral head and neck become more vague. The latter thicken due to new bone formation, while the angle of its sagittal axis to the anatomical femoral axis changes (Figure 14).2,38
 For the frog leg ventrodorsal radiograph of pelvis, the animal’s position is the same as the previous one, with the difference that the hips are abducted in such a way that the femurs are perpendicular to the spine, while the other joints of the hind limbs rest in their normal position without being held (Figure 15). Several degenerative changes are evaluated with this projection and mainly include the presence of osteophytes and/or hypertrophic round ligament of the femur in the acetabular cavity. In the presence of these tissues, total coverage of the femoral head by the acetabulum is prevented as confirmed by the increase in distance between them.27,38
For the frog leg ventrodorsal radiograph of pelvis, the animal’s position is the same as the previous one, with the difference that the hips are abducted in such a way that the femurs are perpendicular to the spine, while the other joints of the hind limbs rest in their normal position without being held (Figure 15). Several degenerative changes are evaluated with this projection and mainly include the presence of osteophytes and/or hypertrophic round ligament of the femur in the acetabular cavity. In the presence of these tissues, total coverage of the femoral head by the acetabulum is prevented as confirmed by the increase in distance between them.27,38
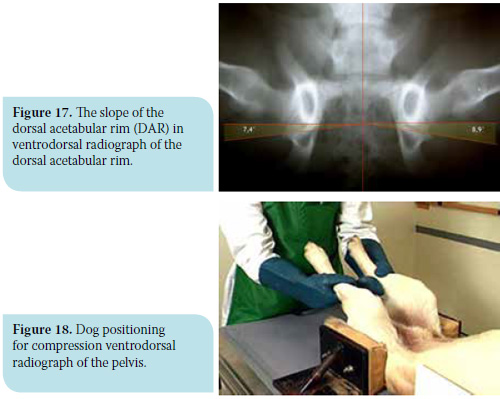
The dorsoventral projection of the dorsal acetabular rim is performed with the animal in sternal recumbency and the hind limbs are pulled cranially and in contact with the lateral thoracic wall of the same side (Figure 16). This projection is useful for the calculation of the slope of the dorsal acetabular rim (DAR). In normal animals, the dorsal acetabular rim is clearly outlined, forming an acute angle. The angle formed by the straight line from the intrarticular part of the dorsal acetabular rim to the sagittal axis of the pelvis and the vertical line on the point the previous lines meet is defined as the dorsal acetabular rim slope and should normally be less than 7.5o (Figure 17). In animals with HD, the projection of the angle of the dorsal acetabular rim is obtuse and rounded due to deterioration, and the slope may even exceed 20o. It should be mentioned that with this projection, the presence of osteophytes on the dorsal acetabular rim may be observed.27
For the lateral projection of the pelvis, the animal is lain on its side. This facilitates assessment of the lumbosacral part of the spine, and enables differentiation of HD from other diseases, such as discospondylitis, spondyloarthritis and disk prolapse, as well as deteriorative lumbosacral stenosis with or without vertebrae bridging (L7-CX1), which is often related to the rather painful cauda equina syndrome.27
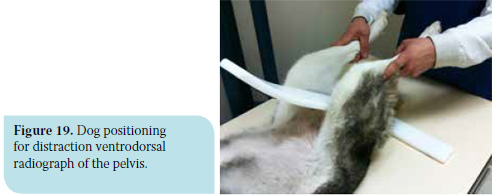
 The compression ventrodorsal radiograph of the pelvis requires the animal to be positioned in dorsal recumbency, the stifles flexed and the femurs almost perpendicular to the table. With the use of special levers (Figure 18), mild adduction force is applied to the stifles, so as to push each femoral head into the respective acetabulum. Measurable variables are obtained by this radiograph as concerns the contact of the articular surfaces and the presence of osteophytes in the acetabulum cavity. The Compression Index (CI) is measured with this projection and is calculated by the ratio of the distance between the geometrical centre of the femoral head and the curving centre of the respective acetabulum to the length of the femoral head radius. More specifically, if there is material in the acetabulum preventing perfect contact with the femoral head, the projectional centre of the femoral head with the curving centre of the acetabulum does not coincide. This material consists of either hypertrophic round ligament implying joint laxity, or osteophytes indicating osteoarthritis. In a normal joint, the compression index equals zero.39,40
The compression ventrodorsal radiograph of the pelvis requires the animal to be positioned in dorsal recumbency, the stifles flexed and the femurs almost perpendicular to the table. With the use of special levers (Figure 18), mild adduction force is applied to the stifles, so as to push each femoral head into the respective acetabulum. Measurable variables are obtained by this radiograph as concerns the contact of the articular surfaces and the presence of osteophytes in the acetabulum cavity. The Compression Index (CI) is measured with this projection and is calculated by the ratio of the distance between the geometrical centre of the femoral head and the curving centre of the respective acetabulum to the length of the femoral head radius. More specifically, if there is material in the acetabulum preventing perfect contact with the femoral head, the projectional centre of the femoral head with the curving centre of the acetabulum does not coincide. This material consists of either hypertrophic round ligament implying joint laxity, or osteophytes indicating osteoarthritis. In a normal joint, the compression index equals zero.39,40
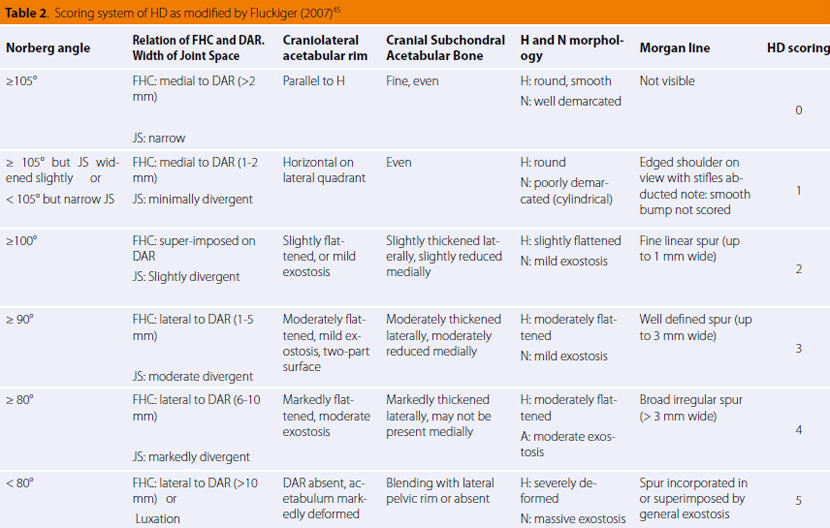
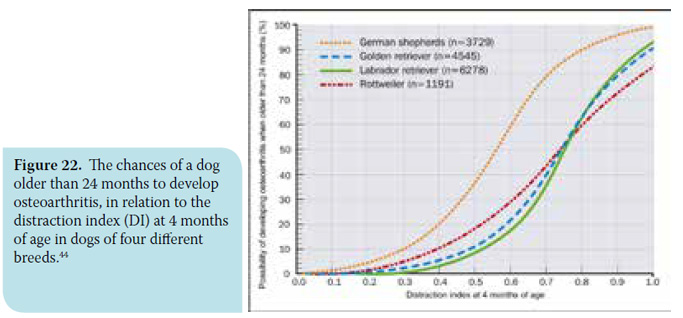
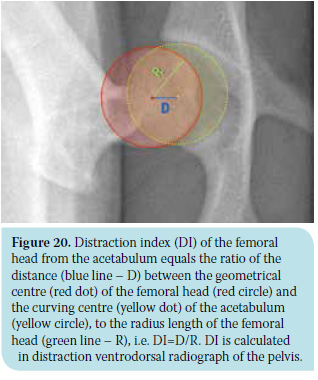 The distraction ventrodorsal radiograph of the pelvis is performed with the animal positioned in the same way as previous, with the difference that a special device is placed between the hind limbs (Figure 19). This works like a lever, pulling the femoral head out of the acetabulum in an outward direction, while the examiner applies mild adduction force to the stifles. The Distraction Index (DI) is calculated with this projection, which is calculated in the same way as the the previous projection (Figure 20). The range of the distraction index varies between 0 (perfect contact of the articular surfaces) and 1 (perfect luxation), while values DI<0.3 are considered physiological. In this way, the laxity grade of the hip can be objectively calculated. The greatest advantage of this method is the fact that it can be performed early in very young animals (4 months of age), and provide useful information in terms of prognosis and consequential therapeutic plan. However, as far as prognosis is concerned, it should be mentioned that although hip laxity predisposes to secondary deterioration, the frequency differs between the several dog breeds. For example, for German Shepherd Dogs with DI >0.3, the chances of developing osteoarthritis are five times greater than those that apply to Rottweiler or Labrador breeds with the same distraction index.2,39-44
The distraction ventrodorsal radiograph of the pelvis is performed with the animal positioned in the same way as previous, with the difference that a special device is placed between the hind limbs (Figure 19). This works like a lever, pulling the femoral head out of the acetabulum in an outward direction, while the examiner applies mild adduction force to the stifles. The Distraction Index (DI) is calculated with this projection, which is calculated in the same way as the the previous projection (Figure 20). The range of the distraction index varies between 0 (perfect contact of the articular surfaces) and 1 (perfect luxation), while values DI<0.3 are considered physiological. In this way, the laxity grade of the hip can be objectively calculated. The greatest advantage of this method is the fact that it can be performed early in very young animals (4 months of age), and provide useful information in terms of prognosis and consequential therapeutic plan. However, as far as prognosis is concerned, it should be mentioned that although hip laxity predisposes to secondary deterioration, the frequency differs between the several dog breeds. For example, for German Shepherd Dogs with DI >0.3, the chances of developing osteoarthritis are five times greater than those that apply to Rottweiler or Labrador breeds with the same distraction index.2,39-44
> Classification systems of HD
The need for early HD diagnosis even in its milder forms, which will allow precautionary measures to be taken and assist selective breeding, has led to the development of several classification systems based on radiographic findings. Mainly four systems are internationally used, as provided by the following institutions:
- FCI (Fédération Cynologique Internationale),
- BVA/KC (British Veterinary Association/The Kennel Club),
- OFA (Orthopedic Foundation for Animals) (www. offa.org) and
- Pennsylvania University of the USA (www.pennhip.org).

The FCI evaluation system has been adopted by at least 80 countries that include the majority of European countries, among which are Greece and Russia, as well as some South American and Asian countries. The scientific committee of FCI proposes a five-grade scoring system ranging from A which reflects a normal hip joint to E indicating severe hip dysplasia (Table 1). Radiographs of the dogs should be made at the age of 1-2 years in order to evaluate the dysplasia grade. The projection used is the ventrodorsal projection of the pelvis with the hind limbs extended, as described above. Scoring is based on the “worse” of the two joints and the variables evaluated include the Norberg angle, subluxation degree of the hip, shape and thickness of the subcortical bone of the cranial acetabulum and femoral neck, shape and size of the craniolateral acetabular rim (Figure 21), and possible evidence of secondary osteoarthritis of the femur in the region where the synovial membrane anchors.45
Unfortunately, evaluation according to this system is not objective because of the high variation between evaluators. This is the reason why many countries like Switzerland, which had first adopted this system, now use several modifications such as the more “quantitative” system of Fluckinger (Table 2).45
The BVA/KC system, which is adopted in Great Britain, Ireland, Australia and New Zealand, is based on nine radiographic criteria evaluated by three radiologists:
1. The Norberg angle
2. The subluxation grade of the hip
3. The cranial acetabular rim
4. The dorsal acetabular rim
5. The craniolateral acetabular rim projection
6. The acetabular fossa
7. The caudal acetabular rim
8. Exostoses on the femoral head and neck
9. The recontouring of the femoral head
Evaluation of the hips is performed in animals older than one year of age. The radiograph used is the ventrodorsal projection of the pelvis with the limbs extended. All the criteria are scored on a scale of 0 and 6, apart from the caudal acetabular rim for which the highest score is 5. The score of the right and left hip are added together in order to obtain the total hip score. The higher the score, the worse the condition of the hips.45
The OFA classification system is mainly used in the USA and Canada. Evaluation is performed by three radiology diplomates. The dog should be older than 24 months in order to be examined and receive certification that no HD is present. Nevertheless, a prime evaluation can be performed right after four months of age. Similar to previous systems, the projection used is the ventrodorsal projection of the pelvis with the limbs extended. Special interest is paid to the following anatomical regions (Figure 21):
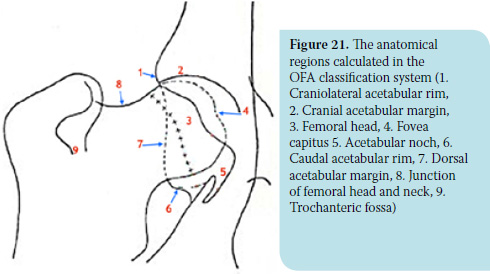 1. Craniolateral acetabular rim
1. Craniolateral acetabular rim
2. Cranial acetabular margin
3. Femoral head (hip ball)
4. Fovea capitus (normal flattened area on hip ball)
5. Acetabular noch
6. Caudal acetabular rim
7. Dorsal acetabular margin
8. Junction of femoral head and neck
9. Trochanteric fossa
The diagnostic criteria are the subluxation grade and degenerative changes of the coxofemoral joint. Based on these criteria, the animals are classified into seven categories and display the following:
a) Normal hips: excellent, good and fair hip categories
b) Dysplastic hips: mild hip dysplasia, moderate hip dysplasia and severe hip dysplasia categories
c) Border line hip dysplasia: evaluation is repeated after six months to be finally categorised as moderate or mild hip dysplasia.45
Penn-HIP (Pennsylvania Hip Improvement Program) is a HD evaluation system, with the use of which both the dysplasia grade and laxity grade of the coxofemoral joint are assessed. This system can be used as early as at four months of age, while three different radiographic projections are used:
a) Ventrodorsal hip-extended radiograph of pelvis
b) Compression ventrodorsal radiograph of the pelvisc)
c)Distraction ventrodorsal radiograph of the pelvis
The coxofemoral joint is evaluated according to qualitative and quantitative data. Qualitative evaluation is related to the shape of the joint and the possible presence of deteriorative changes and is assessed with projection (a). Qualitative evaluation involves calculation of the compression index (CI) at projection (b) and the laxity grade of the joint with calculation of distraction index (DI) at projection (c). As a result, subjective evaluation of other systems can be limited with the use of Penn/HIP, which provides a more objective and proportional model and emphasizes the early evaluation of future osteoarthritis appearance in order to assist selective breeding, precaution in animal management, and possible planning of therapy (Figure 22).45
It is not necessary to perform all the aforementioned imaging techniques during the process of HD diagnosis. If the practitioner is aware of the diagnostic value of each, the appropriate modality can be chosen according to his experience and availability of equipment. Whatever the situation, the differences in disease expression and findings in relation to the animal’s age should not be neglected.
> References
1. Henricson B, Norberg I, Olsson SE. On the etiology and pathogenesis of hip dysplasia: a comparative review. J Small Anim Pract 1966, 7: 673-688.
2. Smith KG, Karbe GT, Agnello KA, McDonald-Lynch MB. Pathogenesis, diagnosis, and control of canine hip dysplasia. In: Vererinary Surgery Small Animal. Tobias KM, Johnston AS (eds). Elsevier Saunders: St. Louis, 2012, pp. 824-848.
3. Mansson J, Norberg I. Dysplasia of the hip in dogs: hormonally induced flaccidity of the ligaments followed by dysplasia of the acetabulum, in puppies. J Small Anim Pract 1965, 6: 121-126.
4. Riser WH, Shirer JF. Hip dysplasia: coxofemoral abnormalities in neonatal German Shepherd dogs. J Small Anim Pract 1966, 7: 7-12.
5. Riser WH, Miller HH. Canine Hip Dysplasia and How to Control it. Orthopedic Foundation for Animals: Philadelphia, 1966.
6. Strayer LM Jr. The embryology of the human hip joint. Yale J Biol Med 1943, 16: 13-26.
7. Strayer LM Jr. Embryology of the human hip joint. Clin Orthop Relat Res 1971, 74: 221-240.
8. Olsson SE. Hoftledsdysplasin pa tillbakagang. Hundsport: Svenska Kennelklubbens Tidskr 1963, 11: 16.
9. Riser WH, Rhodes WH, Newton CD. Hip dysplasia: theories of pathogenesis. In: Textbook of Small Animal Orthopedics. Newton CD (ed). JB Lippincott: Philadelphia, 1985, pp. 953-980.
10. Riser WH. The dog as a model for the study of hip dysplasia: growth, form, and development of the normal and dysplastic hip joint. Vet Pathol 1975, 12: 235-334.
11. Burton-Wurster N, Farese JP, Todhunter RJ, Lust G. Sitespecific variation in femoral head cartilage composition in dogs at high and low risk for development of osteoarthritis: insights into cartilage degeneration. Osteoarthr Cartil 1999, 7: 486-497.
12. Lust G, Summers BA. Early, asymptomatic stage of degenerative joint disease in canine hip joints. Am J Vet Res 1981, 42: 1849-1855.
13. Smith GK, Biery DN, Gregor TP. New concepts of coxofemoral joint stability and the development of a clinical stress-radiographic method for quantitating hip joint laxity in the dog. J Am Vet Med Assoc 1990, 196: 59-70.
14. Morgan JP, Wind A, Davidson AP. Hip dysplasia In: Hereditary Bone and Joint Diseases in the Dog. Schlutersche: Hannover, 2000, pp. 109-203.
15. Madsen JS, Reimann I, Svalastoga E. Delayed ossification of the femoral head in dogs with hip dysplasia. J Small Anim Pract 1991, 32: 351-354.
16. Todhunter RJ, Lust G. Hip dysplasia: Pathogenesis. In: Textbook of Small Animal Surgery. Slatter DH (ed). WB Saunders: Philadelphia, 2003, pp. 2009-2019.
17. Hazewinkel HAW, Goedegebuure SA, Poulos PW, Wolvekamp WC. Influences of chronic calcium excess on the skeletal development of growing Great Danes. J Am Anim Hosp Assoc 1985, 21: 377-391.
18. Hazewinkel HAW. Calcium metabolism and skeletal development in dogs. In: Nutrition of the Dog and Cat. Burger IH, Rivers JPW (eds). Waltham symposium No 7, ed 1. Cambridge University Press: Cambridge, UK, 1989, pp. 293-302.
19. Riser WH, Shirer JF. Correlation between canine hip dysplasia and pelvic muscle mass: a study of 95 dogs. Am J Vet Res 1967, 28: 769-777.
20. Smith GK, Lawler DF, Biery DN, Powers MY, Karbe GT, Shofer F, Gregor TP, Evans RH, Kealy RD. Comparison of primary osteoarthritis of the hip with the secondary osteoarthritis of canine hip dysplasia. In: Proceedings of the 36th Annual Conference of the Veterinary Orthopedic Society. Steamboat Springs, CO, USA, 2009, p. 23.
21. Johnston JD, Noble PC, Hurwitz DE, Andriacchi TP. Biomechanics of the hip. In: The Adult Hip. Callaghan J (ed), 2nd edn. Lippincott Williams & Wilkins: Philadelphia, 2007, pp. 81-90.
22. Powers MY, Martinez SA, Lincoln JD, Temple CJ, Arnaiz A. Prevalence of cranial cruciate ligament rupture in a population of dogs with lameness previously attributed to hip dysplasia: 369 cases (1994–2003). J Am Vet Med Assoc 2005, 227: 1109-1111.
23. Δεσίρης Α, Πατσίκας Μ. Ακτινολογική διερεύνηση της δυ- σπλασίας του ισχίου στο σκύλο. Κτηνιατρική Ενημέρωση 1996, 21: 21-31.
24. Smith GK, Paster ER, Powers MY, Lawler DF, Biery DN, Shofer FS, McKelvie PJ, Kealy RD. Lifelong diet restriction and radiographic evidence of osteoarthritis of the hip joint in dogs. J Am Vet Med Assoc 2006, 229: 690-693.
25. Barr AR, Denny HR, Gibbs C. Clinical hip dysplasia in growing dogs: the long- term results of conservative management. J Small Anim Pract 1987, 28: 243-252.
26. Coopman F, Verhoeven G, Saunders J, Duchateau L, van Bree H. Prevalence of hip dysplasia, elbow dysplasia and humeral head osteochondrosis in dog breeds in Belgium. Vet Rec 2008, 163: 654-658.
27. Slocum B, Slocum TD. Hip. In: Current Techniques in Small Animal Surgery. Bojrab MJ (ed). Williams & Wilkins: Baltimore, 1998, pp. 1127-1185.
28. Roush KJ. Surgical therapy of canine hip dysplasia. In: Vererinary Surgery Small Animal. Tobias KM, Johnston AS (eds). Elsevier Saunders: St. Louis, 2012, pp. 849-864
29. Ortolani M. Congenital hip dysplasia in the light of early and very early diagnosis. Clin Orthop Relat Res 1976, 119: 6-10.
30. Barlow TG. Early diagnosis and treatment of congenital dislocation of the hip. J Bone Joint Surg Br 1962, 44: 292-301.
31. Bardens JW, Hardwick H. New observations on the diagnosis and cause of hip dysplasia. Vet Med Small Anim Clin 1968, 63: 238-245.
32. Bardens JW. Palpation for the detection of joint laxity. In: Proceedings of the Canine Hip Dysplasia Symposium and Workshop. Orthopedic Foundation for Animals: St. Louis, USA, 1972, pp. 105-109.
33. Duerr MF. Hip dysplasia: diagnosis and treatment of the juvenile patient. In: Proceedings of the North American Veterinary Conferrence. Orlando, USA, 2013, pp. 19-23.
34. Jessen CR, Spurrell FA. Radiographic detection of canine hip dysplasia in known age groups. In: Proceedings of the Canine Hip Dysplasia Symposium and Workshop. Orthopedic Foundation for Animals: St Louis, USA 1972, pp. 93-100.
35. Wood JLN, Lakhani KH. Hip dysplasia in Labrador retrievers: the effects of age at scoring. Vet Rec, 2003, 152: 37-40.
36. Vezzoni A, Dravelli G, Vezzoni L, De Lorenzi M, Corbari A, Cirla A, Nassuato C, Tranquillo V. Comparison of conservative management and juvenile pubic symphysiodesis in the early treatment of canine hip dysplasia. Vet Comp Orthop Traumatol 2008, 21: 267–279.
37. Piermattei DL, Flo GL, DeCamp CE. The hip joint In: Brinker, Piermattei, and Flo’s Handbook of Small Animal Orthopedics and Fracture Repair, 4th ed. Saunders Elsevier: St. Louis, 2006, pp. 461- 511.
38. Mauragis D, Reese JD, Berry RC. Small animal pelvic radiography. Today’s Veterinary Practice 2012, 2: 48-53.
39. Smith GK, Biery DN, Gregor TP. New concepts of coxofemoral joint stability and the development of a clinical stress-radiographic method for quantitating hip joint laxity in the dog. J Am Vet Med Assoc 1990, 196: 59-70.
40. Heyman SJ, Smith GK, Cofone MA. Biomechanical study of the effect of coxofemoral positioning on passive hip joint laxity in dogs. Am J Vet Res 1993, 54: 210-215.
41. Mayhew PD, McKelvie PJ, Biery DN, Shofer FS, Smith GK. Evaluation of a radiographic caudolateral curvilinear osteophyte on the femoral neck and its relationship to degenerative joint disease and distraction index in dogs. J Am Vet Med Assoc 2002, 220: 472-476.
42. Runge JJ, Kell, SP, Gregor TP, Kotwal S, Smith GK. Distraction index as a risk factor for osteoarthritis associated with hip dysplasia in four large dog breeds. J Small Anim Pract 2010, 51: 264-269.
43. Smith GK, Gregor TP, Rhodes WH, Biery DN. Coxofemoral joint laxity from distraction radiography and it’s contemporaneous and prospective correlation with laxity, subjective score, and evidence of degenerative joint disease from conventional hipextended radiography in dogs. Am J Vet Res 1993, 54: 1021-1042.
44. Smith GK, Mayhew PD, Kapatkin AS, McKelvie PJ, Shofer FS, Gregor TP. Evaluation of risk factors for degenerative joint disease associated with hip dysplasia in German Shepherd dogs, Golden Retrievers, Labrador Retrievers, and Rottweilers. J Am Vet Med Assoc 2001, 219: 1719-1724.
45. Flückiger M. Scoring radiographs for canine hip dysplasia - the big three organizations in the world. EJCAP 2007, 17: 135–140.



