Retrospective study of five cases
> Abstract
Equine lymphoedema, a common pathologic condition characterized by lymph stasis, requires immediate treatment to ensure a positive outcome. Ascertaining the specific causative agent is not always possible. Despite typical signs of the syndrome, the complex pathogenesis impedes the establishment of an aetiologic treatment protocol. The use of flavonoids in human lymphoedema in recent years has provided the basis for their experimental use in equine lymphoedema. In this study, a treatment protocol for the management of lymphoedema was applied to five horses. The treatment, which included conservative measures, anti-inflammatory and antimicrobial therapy as well as flavoinoids, provided encouraging results, indicating that flavonoids could be a promising therapeutic option for the treatment of equine lymphoedema.
> Introduction
Lymphoedema is characterized by lymphatic stasis. Increased hydrostatic blood pressure with a simultaneous decrease in osmotic blood pressure may lead to the development of subcutaneous oedema, inflammation and plasma protein extravasation. A defence mechanism is provided at excessive protein level through the development of collagen fibres in the subcutaneous tissue which subsequently leads to fibrosis and disruption of the lymphatic and blood circulation. Lymphatic vessels undergo degenerative changes, including disruption of their elastic fibres, progressively causing lymphatic vascular failure.1,2,3
Clinical symptoms of the syndrome may vary and include oedema and different degrees of lameness in one or more limbs, depression, and other type-specific symptoms such as skin lesions, serous or purulent exudate, or even the involvement of the respiratory system.
Clinical and aetiologic diagnosis is based on the history and typical signs while final diagnosis is only reached after microscopic examination of lesion scrapings, culture and histopathology. The management of equine lymphoedema involves both conservative and medical treatment. Lymphoedema requires immediate therapeutic intervention in order to prevent permanent lesions and relapses. In order to achieve a positive outcome, equine lymphoedema should be treated as a serious medical condition requiring immediate and aggressive treatment.4
The purpose of this study is to present five cases with equine lymphoedema that were admitted to the Equine Unit, Faculty of Veterinary Medicine, Aristotle University of Thessaloniki during the period 2010-2011. Ιn conjunction with the standard treatment protocol that usually leads to permanent oedema, flavonoids were used for the first time with encouraging results.
> Materials and Methods
This study included five Warmblood geldings, aged 9 to 14 years old (median = 11 years), with clinical signs of lymphoedema. Prior to each animal’s inclusion in this clinical study, the owner’s consent was obtained.
All horses presented with an acute onset of the syndrome. Physical examination revealed severe diffuse swelling of a hindlimb and elevated regional skin temperature. Lesions usually extended from the pastern proximally to the upper level of the metatarsal region (3/5) and rarely up to the middle of the tibia (2/5) (Figure 1). In all cases, a moderate degree of lameness was detected (2-3/5).
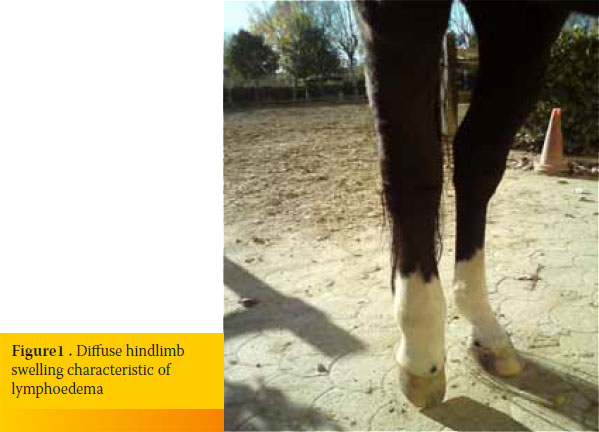
Initial therapy included conservative measures in conjunction with medical treatment. In all horses, conservative treatment included stall confinement, cold hosing three times per day for 15 minutes or astringent agents, such as clay or ice, for five days. Special attention was given to reduce the daily carbohydrate and protein intake by excluding grain and high protein forage, such as alfa-alfa hay, from the daily diet. Massage with cooling gel (Keratex Cooling Gel®) that started distally and moved proximally, in conjunction with pressure bandaging, enhanced lymphatic circulation.
Medical treatment included NSAIDS (phenylbytazone 2.2 mg/kg i.v./24h for seven days), diuretics (furosemide 1mg/kg i.m./24h for five days), antimicrobial agents (oxytetracycline 5-10 mg/kg i.m./24h in two horses, marbofloxacin 1-2 mg/kg iv/24h in three horses for six days) and DMSO (0.5 gr/kg i.v. every 48 hours for three times in total). After the first seven days, all cases were treated with flavonoids (Daflon® 5 mg/ kg every 12 hours per os) for 10 days.
> Results
In 4 cases, initial treatment resulted in 50% reduction of lymphoedema during the first seven days and almost complete elimination by the end of the treatment with flavonoids. In one case no improvement was detected within the first seven days, and Daflon® led to 30% reduction of lymphoedema.
> Discussion- Conclusions
In 4 cases, initial treatment resulted in 50% reduction of lymphoedema during the first seven days and almost complete elimination by the end of the treatment with flavonoids. In one case no improvement was detected within the first seven days, and Daflon® led to 30% reduction of lymphoedema.The lymphatic system, which is a subset of the circulatory system, collects part of the lymph, which is subsequently drained through efferent lymphatic vessels into venous circulation.5
The lymphatic system has many functions, including the removal of various substances and cells from connective tissue. Two main forces control fluid exchange in connective tissue; blood hydrostatic pressure which supplies the connective tissue with nutrients through the capillaries and osmotic pressure which depends mainly on the concentration of albumin and promotes the transport of fluid from the interstitium to blood stream. The main function of the lymphatic system is the removal of metabolic products in order to maintain tissue homeostasis. Lymphatic capillaries, connected to a diffuse network that runs throughout connective tissue, are responsible for the recruitment of high molecular substances and associated liquid compounds.6,7,8
Lymphoedema or lymphatic failure is the insufficiency of lymphatic system to relieve the protein overload that cannot be removed through any other route. There are two main types of lymphatic failure: a) Primary lymphatic failure, characterized not by protein overload but by the inability of lymphatic system to drain the lymph (primary lymphoedema), and b) secondary lymphatic failure, characterized by a normal lymphatic system which is been overloaded by proteins (venous oedema),8,9,10 resulting in lymphatic insufficiency. Disruption or stasis of lymphatic circulation results in the accumulation of substances that can only be removed through lymphatic drainage in the connective tissue. The accumulated proteins retain water. The final outcome, called lymphedema, maintaining high protein levels, promotes the production of collagen fibers (fibrosis) and inflammation. Oedema results in hyperplastic fibrosis and inflammation, a common manifestation of many clinical syndromes and a pathognomonic physical finding of lymphoedema. When the lymphatic system is overloaded, oedema is initially low in proteins; however, progressive overload reduces the ability of the lymphatic system to cope with the ever changing demands as is unable to activate its adaptive structural and functional mechanisms. Dynamic failure turns into mechanical failure.10,11
The exact causes of equine lymphoedema are not fully understood. Several factors are implicated, such as high-protein diet, dehydration, prolonged box rest, high energy diet, lack of exercise, pastern dermatitis, or any other cause that can lead to lymph stasis. Lymphoedema may also be idiopathic, like chronic progressive lymphoedema in draft horses. This syndrome has a typical clinical picture which is characterised by an acute onset of symptoms.4 Diffuse oedema with varying degrees of lameness, especially on the rear limbs, is usually noted. In most cases, no lameness is present whereas when detected, it varies from 1/10 to non-weight bearing lameness. Moreover, rare clinical manifestations, such as anorexia and fever, have been sporadically reported.
Diagnosis is based on history, typical clinical picture and ultrasonographic examination. According to owners, the syndrome is characterized by acute onset of symptoms.
Prognosis is relatively poor since frequent relapses may result in the development of connective tissue in the underlying layers, leading to the formation of permanent lesions in the lymphatic system.4
Studies conducted in human patients indicate that in order to limit the development of lymphoedema and minimize permanent functional lesions, it is necessary to diagnose and treat this syndrome in its early stages.11,12 This need for early diagnosis of chronic lymphoedema led to the development of new techniques. In human medicine, the capacity for lymphoscintigraphy to detect the presence of fluid in the interstitial space and reduced lymphatic drainage renders it a primary diagnostic method in patients with suspected lymphoedema.13,14 Over the last decade, it has also been successfully used for early diagnosis of equine lymphoedema.15 In this study, diagnosis was based on the history and typical clinical picture of the affected limb.
A basic conservative approach in human lymphoedema includes external compression bandage and proper limb support, massage in order to facilitate lymphatic drainage, low-level exercise and careful skin care to avoid possible irritation.11,12
A similar therapeutic approach is used in equine lymphoedema; appropriate management measures of high importance include the adjustment of a daily diet based on the horse’s needs and exercise level. Daily regular work is essential, insomuch as the limb can bear weight. Hand walking should take place for ten minutes, three times per day. The application of astringent agents on the affected limbs, such as clay or cooling gel, and daily hosing with cold water two times per day for 15-20 minutes are also important. Massage in a distal-toproximal direction is particularly beneficial. It is also necessary to bandage the affected limbs with special dressings to facilitate lymph circulation.
At this point, a detailed report of the treatment protocol that was used is presented, in addition to management measures.
Phenylbutazone was given at a dose of 2.2 mg/kg once per day as an excellent option for the management of inflammation and pain in horses. Additionally, DMSO was administered at a dose of 0.5 gr/ kg i.v. as a free radical scavenger with strong antiinflammatory activity.3 As an organic liquid solvent, DMSO, among others, has a high water absorbing capacity, contributing to further reduction of oedema. 16,17 The use of furosemide at a dose of 1 mg/kg i.m. once per day for five days was considered advantageous for the treatment of oedema.
As concerns the administration of antibiotics, the use of oxytetracycline (5-10 mg/kg 24h) is reported to have beneficial effects due to its bacteriostatic action against Gram + and Gram- and certain anaerobic bacteria. Oxytetracycline has a dual role in terms of management of lymphoedema; besides its bacteriostatic capacity, it also has the ability to bind calcium causing relaxation of tendons.3 In a similar way, it binds calcium in the wall of the blood vessels causing their relaxation, thereby improving their functionality, as due to dilatation and inflexibility from fibrosis and inflammation they are not working properly.3,18,19
Unfortunately, oxytetracycline has several side effects, particularly when administered intravenously. It should be diluted and administered slowly as rapid intravenous administration can cause collapse of the horse, due to hypotension which is attributed to the rapid binding of calcium with chelates, while some patients may experience an allergic reaction. As an alternative, intramuscular and oral administration may be used. In this study, in order to avoid side effects, oxytetracycline was administered intramuscularly without reactions.19,20
To avoid the aforementioned side effects and because of its broad antibacterial spectrum against Gram + and Gram- bacteria,21,22 marbofloxacin was the antibiotic of choice in 3/5 horses, in cases where cost was an issue.
Non-controlled studies report the use of heparin (40-150 IU/ kg sc) in the treatment of lymphoedema. Heparin may cause bleeding, thrombocytopenia, renal dysfunction, hypokalaemia, alopecia, priapism, while long-term administration may cause osteoporosis.23 Taking into consideration the severe and frequent side effects, heparin was not included in the treatment protocol that was used.
Flavonoids belong to polyphenols and are categorized according to their chemical structure in flavonols, flavones, flavanones, isoflavones, catechins, anthocyanins and chalcones. Over 4,000 flavonoids have been identified, many of which are found in fruits, vegetables and beverages. Mounting interest has been noted in the research into flavonoids in recent years, due to their beneficial effect on human health. Their antiviral, anti-allergic, anti-inflammatory, antineoplastic, antiplatelet and antioxidant activities have been widely reported.24 Moreover, their use in the treatment of phlebitis, lymphoedema, and other vascular disorders is rapidly becoming more widespread.25
In the current study, Daflon® containing 500 mg of purified micronized flavonoid fraction with 90% diosmin (450 mg per tablet) and 10% hesperidin (50 mg per tablet) was administered. This composition is used in humans as adjuvant therapy in the treatment of chronic venous insufficiency of the lower extremities and the symptomatic treatment of haemorrhoidal crisis.26
As previously mentioned, high hydrostatic blood pressure causes inflammation in the valves of superficial blood vessels. Leukocytes adhering to the valves activate inflammatory processes, resulting in disturbance of their normal function (transport of blood and lymph from peripheral tissues). Blood that cannot normally flow back to the heart retrogrades, further increasing blood hydrostatic pressure. The latter, as already mentioned, serves as a triggering factor of lymphoedema.
Diosmin is responsible for the treatment of an inflammatory process. A semisynthetic venotropic drug (modified hesperidin), diosmin prolongs the vasoconstrictor effect of norepinephrine in the venous wall, resulting in increased venous tone and thus reducing venous stasis. Furthermore, diosmin improves lymphatic drainage by increasing the frequency and intensity of the contractions of lymphatic vessels and augmenting the total number of functional lymphatic capillaries.27 Equally important is its ability to reduce the expression of adhesion molecules on the endothelium of blood vessels, thereby inhibiting the adhesion, migration and activation of leukocytes at the capillaries. In consequence, the release of inflammatory mediators, mainly free oxygen radicals and prostaglandins, is inhibited. Despite the undeniable advantages of pure diosmin, it has been reported that the administration of Daflon® (as micronized purified fraction) predominates.28 Additionally, two integrated controlled studies reported that compared with placebo, the use of Daflon® in patients with chronic venous insufficiency led to increased venous tone, reduced venous capacity, while significantly improving the haemodynamic profile.29 Based on the results of clinical studies, the administration of Daflon® resulted in increased lymphatic capillary luminal pressure and diameter, thus facilitating lymph permeability and diffusion, as well as an improvement of the oedema.30,31
In this study, having extrapolated data based on the dosage regimen applied to humans,24-31 we administered Daflon® at a dose of 5 mg/kg BID per os for 10 days; no adverse reactions were observed. All horses maintained a good appetite with no signs of gastrointestinal disease; they showed progressive improvement of oedema and limb function and immediately returned to their previous athletic activity. Even in the one case of lymphoedema that did not respond positively, additional administration of Daflon® to the initial protocol resulted in 30% further reduction of the oedema.
In summary, lymphoedema is a particularly frequent pathological condition that requires immediate and aggressive treatment to prevent the development of irreversible lesions in the affected extremities. The implementation of the protocol described above with the additional use of flavonoids can effectively contribute to the management of this syndrome. It is worth mentioning that this study involved a limited number of horses and no control group so statistically reliable results could not be obtained. The results of this clinical trial indicate that the properties of Daflon®, along with easy administration, the absence of side effects and cost-effective use, make it an excellent ally in the treatment of equine lymphoedema. To validate its full potential, further research is needed.
> References
1. Powell H. Therapy for horses with chronic progressive lymphoedema. Vet Rec 2009, 165: 758.
2. Wollina U, Abdel-Naser MB, Mani R. A review of the microcirculation in skin in patients with chronic venous insufficiency: the problem and the evidence available for therapeutic options. Int J Low Extrem Wounds 2006, 5: 169-180.
3. Rees CA. Disorders of the skin. In: Equine internal medicine. Reed SM, Bayly WM, Sellon DC (eds) 3rd edn. WB Saunders: St Louis, 2004, pp. 627–720.
4. Taylor FGR, Hillyer MH. Lymphatic diseases. In: Diagnostic Techniques in Equine Medicine. WB Saunders: London, 1997, pp. 183-189.
5. Μιχαήλ ΣΓ. Λεμφοφόρο Σύστημα. Σε Συγκριτική Ανατομική των Κατοικίδιων Θηλαστικών. Αδελφοί Κυ- ριακίδη Α.Ε: Αθήνα 2004, σελ. 441-460.
6. Μιχαήλ ΣΓ. Λεμφοφόρο Σύστημα. Ιστολογία (2η έκ- δοση). Αδελφοί Κυριακίδη ΑΕ: Αθήνα 1986, σελ.211-212.
7. Cluzan, RV. Lymphatics and edema. In: Progress in Lymphology. Proceedings of the XIIIth International Congress of Lymphology. Cluzan RV, Pecking AP, Lokiec FM (eds), Excerpta Medica, Elsevier Science, International Congress series: Amsterdam, 1992, pp. 716-717.
8. Tretbar LL. Structure and Function of the Lymphatic System. In: Lymphedema. Springer: London, 2008, pp. 1-11.
9. Pecking A, Cluzan R. Explorations du systeme lymphatique : epreuve au bleu, lymphographies directs, lymphoscintigraphies, autres méthodes. Encycl Med Chir Angéiologie 1997, 19: 1130-1135.
10. Szuba A, Rockson SG. Lymphedema: classification, diagnosis and therapy. Vasc Med 1998, 3: 145-156.
11. Rockson, SG. Lymphedema. Am J Med 2001 110: 288-295.
12. MacLaren, JA. Skin changes in lymphoedema: pathophysiology and management options. Int J Palliat Nurs 2001, 7: 381-388.
13. Patsch H. Assessment of abnormal lymph drainage for the diagnosis of lymphedema by isotopic lymphangiography and by direct lymphography. Clin. Dermatol 1995, 13: 445-450.
14. Moshiri M, Katz DS, Boris M, Yung E. Using lymphoscintigraphy to evaluate suspected lymphoedema of the extremities. Am J Res 2002, 178: 405-412.
15. Cock HEV, Affolter VK, Wisner ER, Larson RF, Ferraro GL. Lymphoscintigraphy of draught horses with chronic progressive lymphoedema. Equine Vet J, 38: 148-151.
16. Brayton CF. Dimethyl sulfoxide (DMSO): a review. The Cornell Veterinarian, 1986, 76: 61-90.
17. Ellson R, Stearns R, Mutz M, Brown C, Browning B, Harris D, Qureshi S, Shieh J, Wold D. In situ DMSO hydration measurements of HTS compound libraries. Com Chem High T Scr 2005, 8: 489-498.
18. Ibsen KH ,Urist MR. Complexes of calcium and magnesium with oxytetracycline. Experimental Biology and Medicine 1962, 109: 797-801.
19. Andersson G, Ekman L, Månsson I, Persson S, Rubarth S, Tufvesson G. Lethal complications following administration of oxytetracycline in the horse. Nordisk veterinärmedicin 1971, 23: 9.
20. White G, Prior SD. Comparative effects of oral administration of trimethoprim/sulphadiazine or oxytetracycline on the faecal flora of horses. Vet Rec 1982, 111: 316-318.
21. Dowling P. Antimicrobial therapy In: Equine clinical pharmacology. Bertone J, Horspool, LJ. (Eds.) Saunders: Philadelphia, 2004, pp. 267-271.
22. Lees P, Aliabadi FS. Marbofloxacin in equine medicine: have we got the doses right? Equine Vet J 2002, 34: 322-325.
23. Φαρμακολογία Μαρία Κουτσοβίτη-Παπαδοπού- λου, Αριστοτέλειο Πανεπιστήμιο Θεσσαλονίκης τμήμα εκδόσεων, Πανεπιστημιακό τυπογραφείο: 2004-2005, σελ.120.
24. Burda S, Oleszek W. Antioxidant and antiradical activities of flavonoids. J Agric Food Chem 2001, 49: 2774-2779.
25. Vettorello G, Cerreta G , Derwish A , Cataldi A, Schettino A, Occhionorelli S, Donini I. Contribution of a combination of alpha and beta benzopyrones, flavonoids and natural terpenes in the treatment of lymphedema of the lower limbs at the 2d stage of the surgical classification. Minerva cardioangiologica 1996, 44: 447-455.
26. Barbe R, Amiel M. Pharmacodynamic properties and therapeutic efficacy of Daflon 500 mg. Phlebology 1992, 7: 41-44
27. Cospite M, Dominici A: Double blind study of the pharmacodynamic and clinical activities of 5682 SE in venous insufficiency. Advantages of the new micronized form. Int Angiol 1989, 8: 61-65.
28. Allegra C, Bartolo Jr M, Carioti B, Cassiani, D. An original microhaemorheological approach to the pharmacological effects of Daflon 500 mg in severe chronic venous insufficiency. Int J Microcirc Clin Exp 1995, 15: 50-54.
29. Ibegbuna V, Nicolaides AN, Sowade O, Leon M, Geroulakos G.Venous elasticity after treatment with Daflon 500 mg. Angiology 1997, 48: 45-49.
30. Struckmann JR. Clinical efficacy of micronized purified flavonoid fraction: an overview. J Vasc Res 1999, 36: 37-41.
31. Cesarone MR, Laurora G, De Sanctis MT, Belcaro, G. Capillary filtration and ankle edema in patients with venous hypertension: effects of Daflon. Angiology 1993, 44: 57-61.



